COVID-19 Vaccine-Associated Transient Amnesia
We observed cases of transient global amnesia (TGA) after the 1st or 3rd dose of a COVID-19 vaccine in the elderly population of our study.
Recent review of reports from the World Health Organization VigiBase® ideitified 289 TGA cases associated with a COVID-19 vaccine. The Pfizer-BioNTech COVID-19 Vaccine, also known as COMIRNATY® or Tozinameran was mostly represented (147, 50.8%), followed by the Oxford/AstraZeneca or ChAdOx1-SAZD1222 (69, 23,8%), Moderna - SPIKEVAX, elasomeran - (60, 20.8%), and JNJ-78436735 (12, 4.2%).
Cerebrovascular, inflammatory, or migrainous mechanisms may underlie this association. Yet, numerous confounding factors cannot be tackled with this approach, and causality cannot be ascertained.
Here is an exemplar medical case:
Two weeks after receiving the first dose of ChAdOx1 nCov-19 vaccine, a disease-free 38-year-old woman experienced acute onset of abnormal symptoms, such as amnesia, incoherent speech, and difficulty typing using communication software.
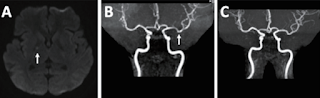
Her immediate adverse reactions following vaccination were only fever and general malaise for 2 days.On arrival at the emergency department, the patient was conscious clear but demonstrated psychomotor slowing and confabulation. Neurological examination revealed high levels of cortical dysfunction including poor judgement; disorientation to time, place, and person; impaired short-term memory; failed abstract thinking; impaired calculation; and impaired language function (including partial incoherence, agraphia, and anomic aphasia). The language domains of fluency, reading, and repetition remained notably intact. No cranial nerve, motor, sensory, or coordination dysfunction were observed. Brain computed tomography revealed negative findings of hemorrhage and structural lesion. While waiting in the emergency room, the patient experienced a first-time seizure with a general pattern of a tonic–clonic seizure. The patient was prescribed lorazepam and levetiracetam, and the seizure subsided. The patient presented postictal confusion and agitation. She was admitted for examination and treatment. Brain MRI revealed a subacute infarction at the right internal capsule through diffusion-weighted imaging (Fig. 1A), and magnetic resonance angiography revealed an irregularity in vascular contour, indicating potential vasculopathy, such as vasculitis (Fig. 1B). EEG revealed diffuse background slowing with intermittent low to moderate voltage sharp transients at the right frontotemporal region.
Qualitative serum tests for paraneoplastic neurologic syndrome, including anti-Tr(DNER), anti-GAD65, antiZic4, anti-titin, anti-SOX-1, anti-Recovertin, anti-Hu, anti-Yo, anti-Ri, anti-PNMA2(Ma2/Ta), anti-CV2, and anti-amphiphsyin, were all negative. Qualitative serum and CSF tests for limbic encephalitis, including antiNMDAR, anti-AMPAR1, anti-AMPAR2, anti-GABABR, anti-LGI1, and anti-CASPR2, were all negative. Although autoantibodies were all absent detected, seronegative autoimmune encephalopathy triggered by the ChAdOx1 nCov-19 vaccination was highly suspected. In addition to levetiracetam, the doctors initiated steroid pulse therapy at the start of treatment. The regimen was methylprednisolone 1000 mg IV daily for 5 days, which was subsequently changed to prednisolone 30 mg PO daily starting from day 6 of admission. The patient’s symptoms dramatically improved from day 3 of admission. The patient was discharged without neurological deficit or sequelae. One week after discharge, the patient received repeat EEG, which revealed resolved slow waves without epileptiform discharges (Fig. 3). Three months later, repeated brain MRI revealed regressive change of previous irregularity of vascular contour. (Fig. 1C)
REFERENCES
Merino D, Gérard AO, Van Obberghen EK, Ben Othman N, Ettore E, Giordana B, Viard D, Rocher F, Destere A, Benoit M, Drici MD. COVID-19 Vaccine-Associated Transient Global Amnesia: A Disproportionality Analysis of the WHO Safety Database. Frontiers in Pharmacology. 2022:1858.
Huang YJ, Huang CS. Postvaccinal Encephalopathy Presenting with Amnesia and Seizure After ChAdOx1 nCov-19 Vaccination: A Case Report. Acta Neurologica Taiwanica. 2022 Sep 18;31(3):111-5.

Comments
Post a Comment