Long VITT
There is an increased rate of coagulation disorders following all COVID-19 vaccines. One example is Vaccine-induced immune thrombotic thrombocytopenia (VITT). It happens when the body's defense mechanisms incorrectly target and kill platelets, small cell fragments that aid in blood clotting. VITT can lead to significant morbidity and mortality due to the various thrombosis-related complications.
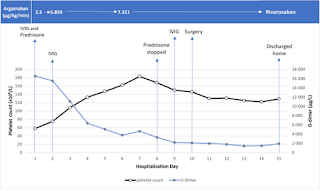
When the Polyspecific Anti-PF4 Immunoassay (Immucor) result was positive, VITT was quickly suspected. The PF4-SRA was also positive. Prednisone, intravenous immunoglobulin, argatroban, and rivaroxaban were used to treat the patient, who also underwent lower limb revascularization surgery. The clinical course was favorable despite PF4-SRA still being positive after successful surgical intervention. After receiving care, the patient was sent home.
After a 2-year follow-up, the patient developed thrombocytopenia and positive platelet-activation tests. He is not known for any relevant past medical history other than alcohol consumption and slight thrombocytopenia (130 × 109/L since 2015). It is unknown if the chronic platelet-activating anti-PF4 antibodies or the patient's habits are to blame for the continuous and progressively serious thrombocytopenia. Another case with recurrent episodes of thrombocytopenia and positive PF4-SRA over a 9-month follow-up period in a 69-year-old male was also reported.
The authors suggested that long VITT diagnosis definition might include the persistence within patient serum/plasma of anti-PF4 platelet-activating antibodies with clinical manifestations (e.g., thrombocytopenia) for more than 3 months.
REFERENCES
Roberge G, Carrier M. Long VITT: A case report. Thromb Res. 2023 Jan 24;223:78-79. doi: 10.1016/j.thromres.2023.01.017. Epub ahead of print. PMID: 36709677; PMCID: PMC9870606.
Roberge G, Côté B, Calabrino A, Gilbert N, Gagnon N. Acute lower limb ischemia caused by vaccine-induced immune thrombotic thrombocytopenia: focus on perioperative considerations for 2 cases. Thrombosis Journal. 2022 Jul 4;20(1):38.
Gabarin N, Arnold DM, Nazy I, Warkentin TE. Treatment of vaccine-induced immune thrombotic thrombocytopenia (VITT). InSeminars in hematology 2022 Mar 7. WB Saunders.
Berild JD, Larsen VB, Thiesson EM, Lehtonen T, Grøsland M, Helgeland J, Wolhlfahrt J, Hansen JV, Palmu AA, Hviid A. Analysis of thromboembolic and thrombocytopenic events after the AZD1222, BNT162b2, and MRNA-1273 covid-19 vaccines in 3 Nordic countries. JAMA Network Open. 2022 Jun 1;5(6):e2217375-.

Comments
Post a Comment