Acute and Long-term post-vaccinal complications
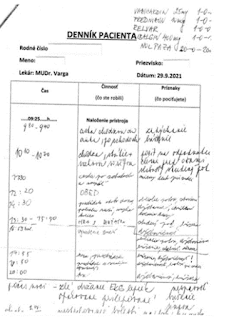
There were no corresponding acute changes seen in the T1 and T2 maps, which are used to assess tissue characteristics. Handwritten note describes polymorphic patient complaints during 24-hour monitoring EKG (Department of Functional Diagnostics, UN - St. Michael's Hospital, a. s.; 29.9.2021)
The patient's condition stabilized and gradually improved with treatment. Five months later, the patient was able to do 5 squats or cycle on a stationary bike for a few minutes without significant symptoms. In an orthostatic test, the patient's tolerance interval for upright posture was prolonged from 2 to 7.5 minutes.
REFERENCE
Varga I, Michalka P, Mištinová JP. Complications after administration of mRNA vaccine against COVID-19 - case report and short review. Vnitr Lek. 2023 Summer;69(E-4):20-27. English. doi: 10.36290/vnl.2023.054. PMID: 37468319.

Comments
Post a Comment